Big deal! Multitude and Adcendo signed a licensing agreement for a ADC drug
On August 20, 2024, Multitude Therapeutics Inc. (“Multitude”) and Adcendo ApS (“Adcendo”) jointly announced that they have signed a licensing agreement for ADCE-T02 (AMT-754), a novel antibody-drug conjugate (ADC) targeting Tissue Factor (TF). Under this agreement, Adcendo would obtain the exclusive development and commercialization rights for the asset globally, except for the Greater China region. Meanwhile, Multitude would receive upfront and milestone payments totally over $1 billion, as well as tiered royalties on future product sales.
ADCE-T02 is designed by Multitude based on its T moiety ADC technology platform. This platform adopts a highly hydrophilic self-cleaving linker modulation technology. By modifying the PABC moiety (T moiety) in the ADC architecture with groups such as m-methylaminomethyl (T800), PEG (T900), or polysarcosine (pSAR, T1000), it achieves a superior hydrophobic shielding effect and stability (Fig. 1).
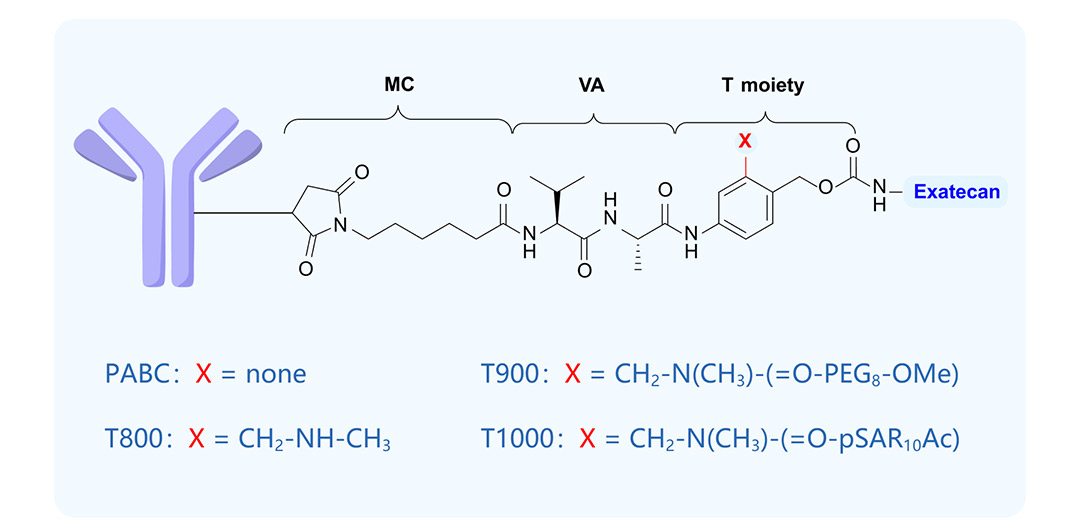
(Fig. 1) Antibody–exatecan conjugate by T moiety
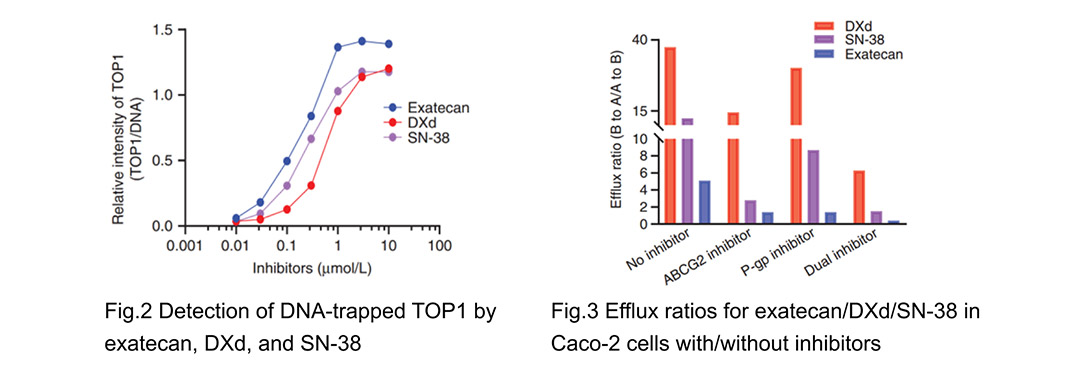
Camptothecin class TOP1 inhibitors have become more prominent as ADC payloads because of the launch of DS-8201a and Trodelvy. Yet, due to tumor heterogeneity and multidrug resistance (MDR) regulation, DXd/SN-38 ADCs face intrinsic and acquired resistance, highlighting the need for more effective ADCs. ADCE-T02 utilizes exatecan as payload owing to its higher TOP1 inhibition potency (Fig. 2), lower sensitivity to multidrug resistance (MDR) (ABCG2/ P-gp) (Fig. 3), higher permeability, bystander penetration and its known clinical and toxicity profile in humans compared to DXd/SN-38. However, exatecan is too hydrophobic to be conjugated directly to antibodies, due to its unique condensed polycyclic structure.
Based on the characteristics of exatecan, Multitude modified the T moiety of ADCE-T02 with polysarcosine (pSAR, T1000) via the T moiety–exatecan technology. This modification overcame the challenge that exatecan is too hydrophobic to be conjugated directly to antibodies, as pSAR exhibits higher solubility and biocompatibility (Fig. 4).
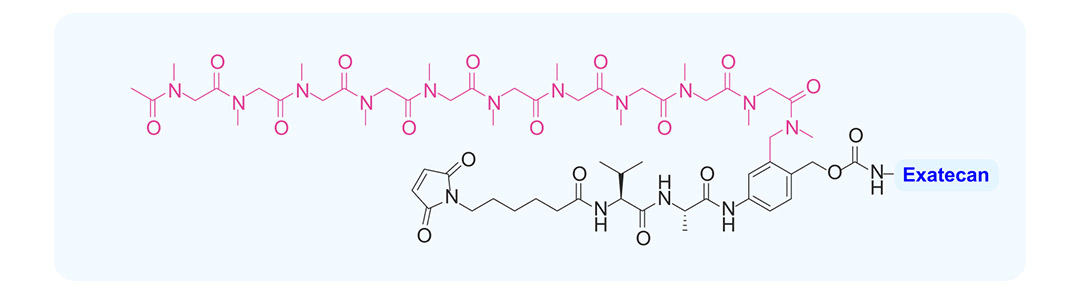
(Fig. 4) Structure of T1000
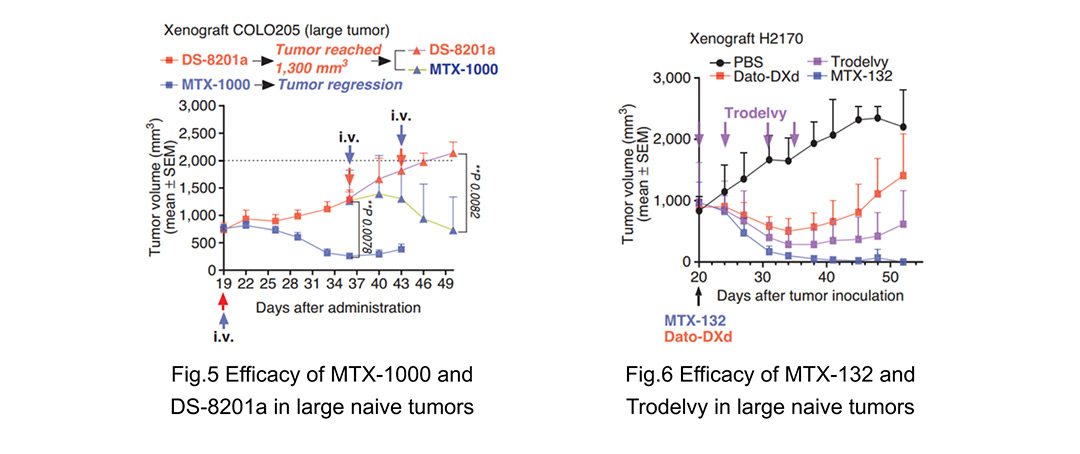
According to studies[1] conducted by Multitude, T moiety–exatecan ADCs were more effective against large-size tumors due to a higher intratumor pharmacodynamic response and were able to overcome MDR+ (high ABCG2/P-gp expression) tumor resistance to DXd/SN-38 ADCs in vivo. Taking MTX-1000 (Tras-T1000-exatecan) (Fig. 5) and MTX-132 (hRS7-T1000-exatecan) (Fig. 6) as examples, In xenograft models of starting tumor size of 500 to 1,000 mm3, MTX-1000 and MTX-132 induced tumor regression and were significantly more effective than DS8201a and Trodelvy, respectively. Furthermore, MTX-1000 induced tumor regression in the DS-8201a–treated group when the tumor reached 1,300 mm3, whereas another dose of DS-8201a failed to inhibit tumor growth. The data indicates that, compared to prior ADCs, T moiety–exatecan ADCs have the potential to overcome resistance mechanisms and demonstrate efficacy in a larger patient population and a broader range of tumors.
Reference:
[1] Cancer Discov (2023) 13 (4): 950–973.